38 label the water molecule
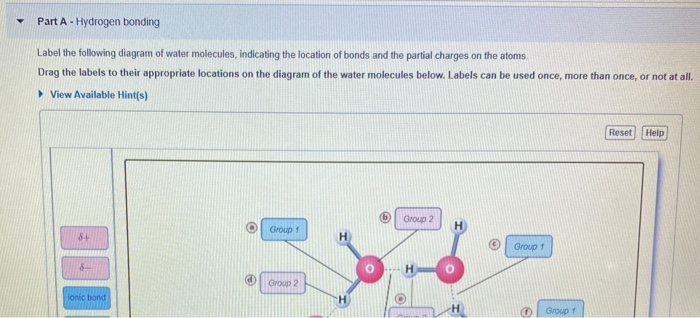
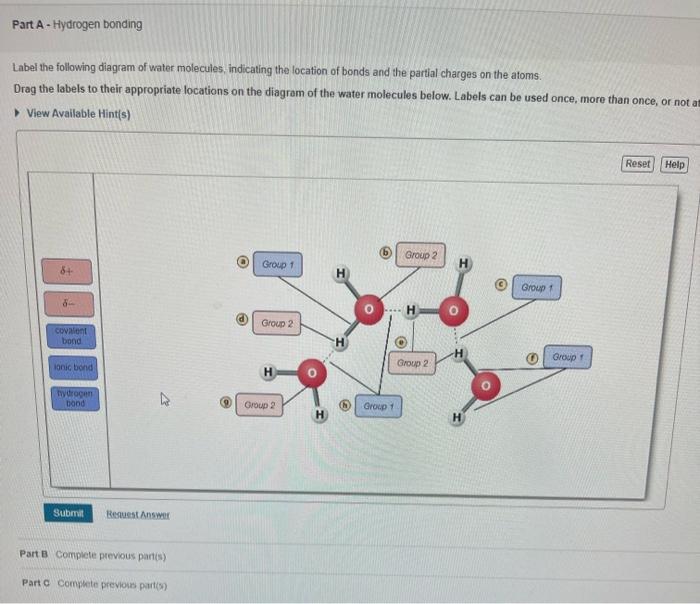
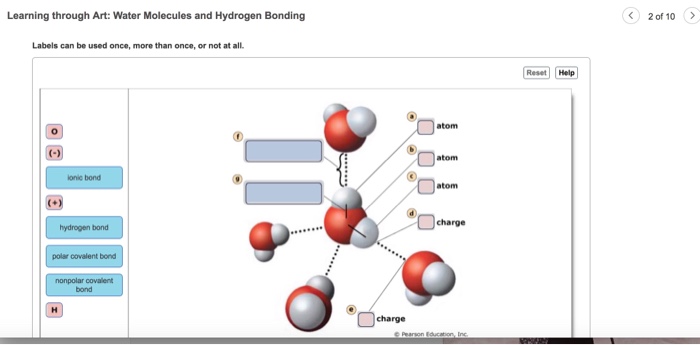
Mastering BSC1010: Chapter 3 Flashcards | Quizlet Label the following diagram of water molecules, indicating the location of bonds and the partial charges on the atoms. Properties of water Classify each statement as an example of adhesion, cohesion, or surface tension Adhesion: Water molecules cling to the side of a breaker. Water molecules cling to plant cell walls. Lesson Worksheet: Water Biology - Nagwa In this worksheet, we will practice describing the structure of water molecules, explaining why water is a polar molecule, and outlining the properties of ...
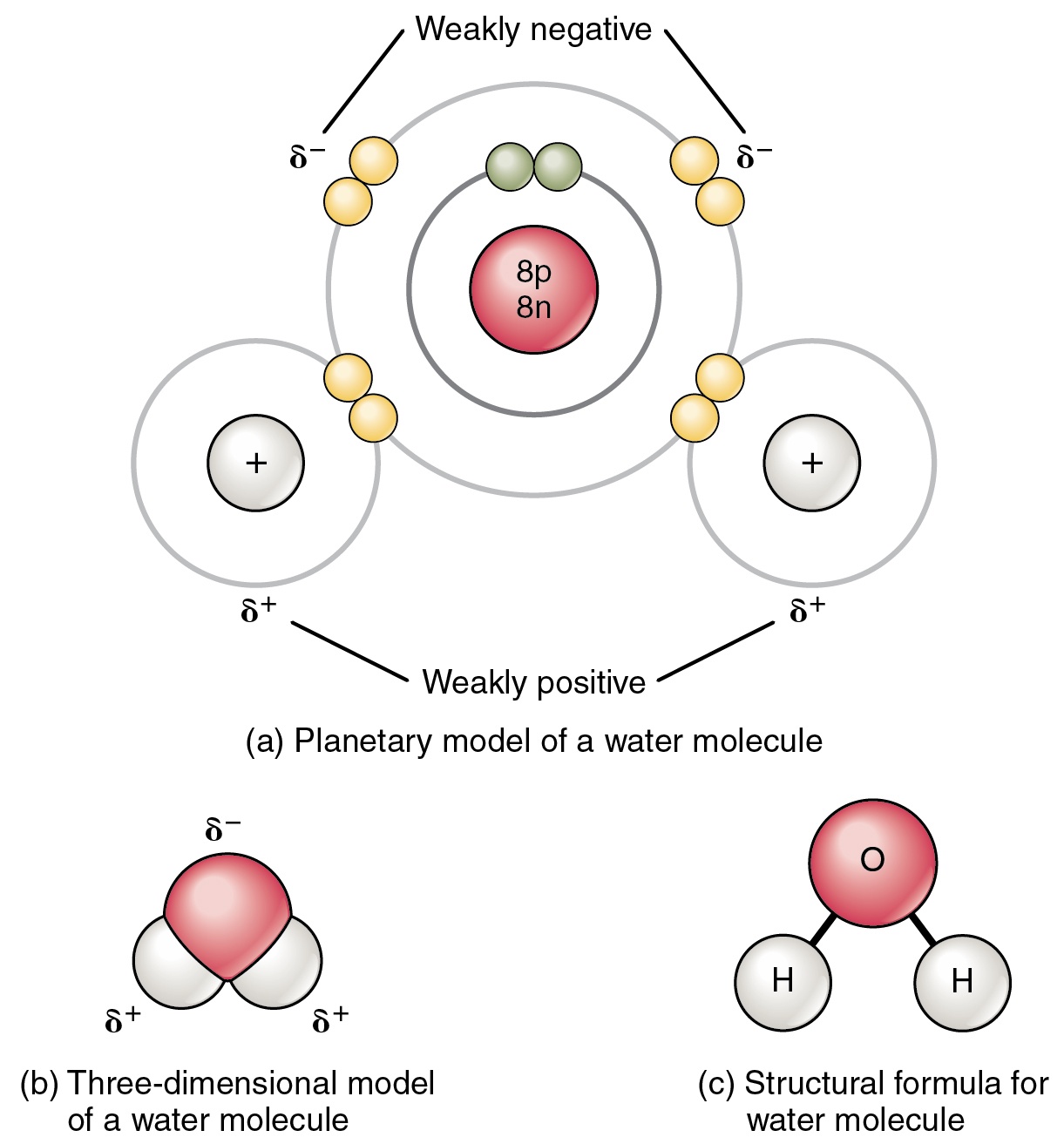
The Configuration of the Water Molecule | EARTH 111: Water: Science and ... A molecule of water is composed of two atoms of hydrogen and one atom of oxygen. The one and only electron ring around the nucleus of each hydrogen atom has only one electron. The negative charge of the electron is balanced by the positive charge of one proton in the hydrogen nucleus.
Label the water molecule
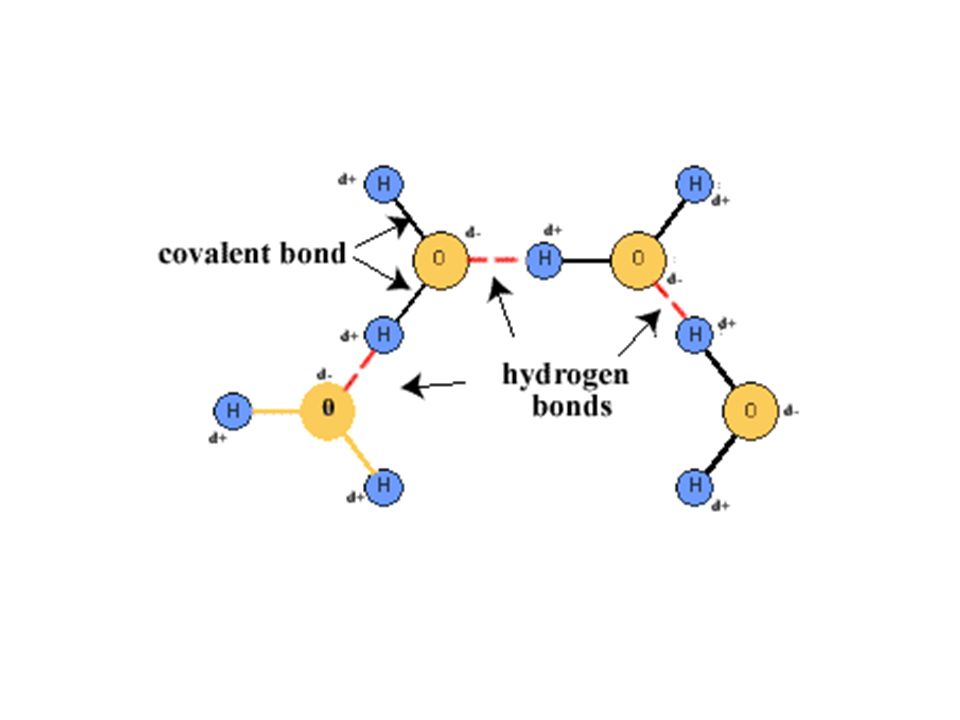
The Structure of Water | Chemistry for Non-Majors | | Course Hero Structure of Water. Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent ( polar bonds ). The oxygen atom attracts the shared electrons of the covalent bonds to a significantly greater extent than the hydrogen atoms. Draw a diagram of water molecules, labeling the hydrogen bond and ... A diagram of water molecules, labeling the hydrogen bond and covalent bond - is attached as an image. Water has hydrogen and oxygen molecules bonded with covalent and, covalent bonds are intramolecular bonds whereas hydrogen bonds are intermolecular bonds. Water is held together with covalent bonds. Draw and label a ball and stick presentation of a water molecule ... This visual representation demonstrates that one atom of oxygen, O, bonds with two atoms of hydrogen, H, to form one molecule of water, ...
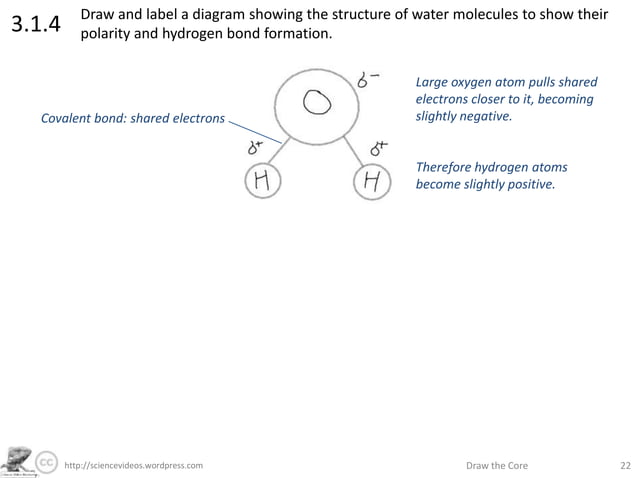
Label the water molecule. 3.1.4 Draw and label a diagram showing the structure of water ... 3.1.4 Draw and label a diagram showing the structure of water molecules Stephanie Castle 22K subscribers Subscribe 76K views 9 years ago OLD SYLLABUS Topic 3: The Chemistry of Life Water... Water: Molecular Structure of Water (With Diagram) - Biology Discussion The Water (H 2 O) molecule has a triangular geometry with O-H bond distance of 0.0965nm and the H-O-H bond angle is 104.5°. Although the water as a whole is electrically neutral, it behaves as an electrical dipole. This is because; oxygen atom is more electronegative than the hydrogen atoms, so it attracts electrons away from the hydrogen nuclei. Solved Label the following diagram of water molecules, - Chegg Science. Biology. Biology questions and answers. Label the following diagram of water molecules, indicating the location of bonds and the partial charges on the atoms Drag the labels to their appropriate locations on the diagram of the water molecules below. Labels can be used once, more than once, or not at all View Available Hint (s) Reset ... Water Molecule - Animation | Prof. Eduardo J. Stefanelli Water molecule - chemistry - animation and text Water Molecule. Colorless, odorless and tasteless, water is one of the most important elements for all known life forms. About 70% of the human body's weight is composed of this molecule. It can be obtained by ingestion of liquids, food or by the combustion of food, as this reaction releases ...
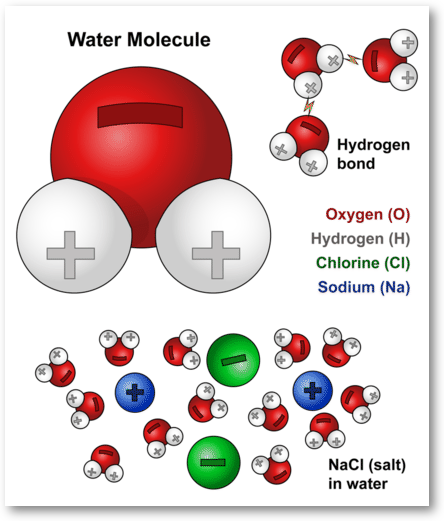
PDF Chapter 3: Water and Life - Pearland High School The fact that water is such a versatile solvent can be traced back to the polarity of the water molecule. Water can dissolve ionic compounds, many compounds made up of nonionic polar molecules, as well as molecules ... 28. On the pH chart, label pH 1-14. Label neutral, acid, and base. Indicate the locations of pure water, urine, gastric juice ... Water molecules and their interaction with salt - USGS Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together. After the salt compounds are pulled apart, the sodium and chloride atoms are surrounded by water molecules, as this diagram shows. Once this happens, the salt is dissolved, resulting in a homogeneous solution. Find out more Adhesion/cohesion Hydrogen bonds in water (article) - Khan Academy A water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. All of the electron pairs—shared and unshared—repel each other. Draw a water molecule and label its polarity (partial positivelpartial ... Apr 28, 2022 ... VIDEO ANSWER: Hello guys. So in this question we have to demonstrate the polarity Of H 20. That is water. So now if you make the structure ...
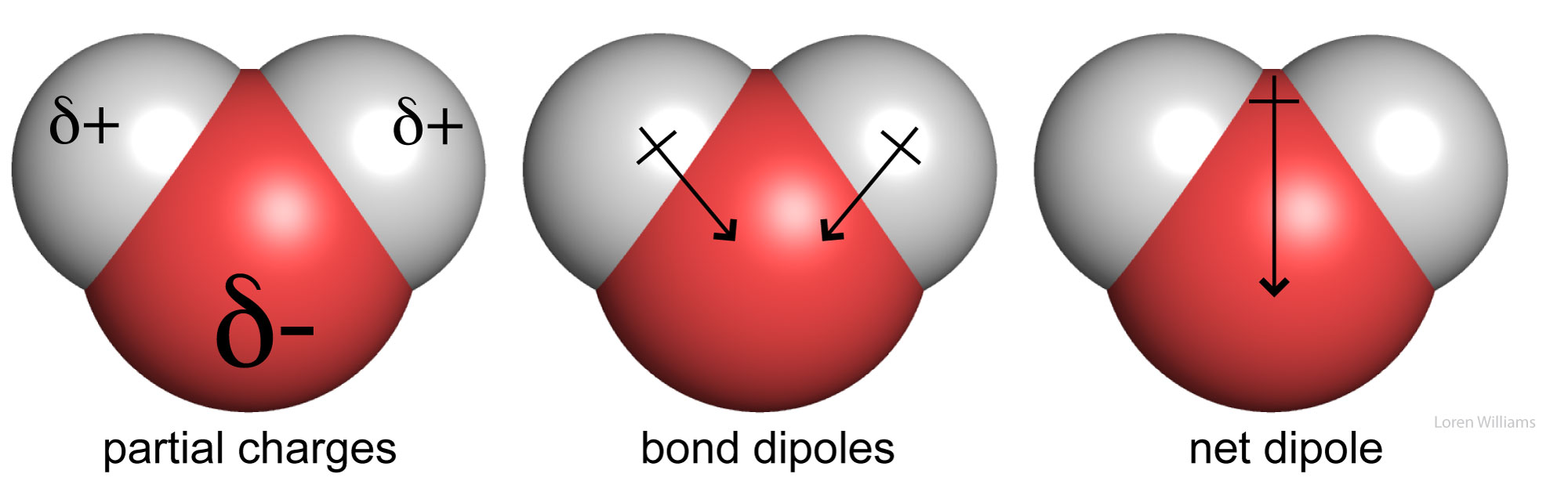
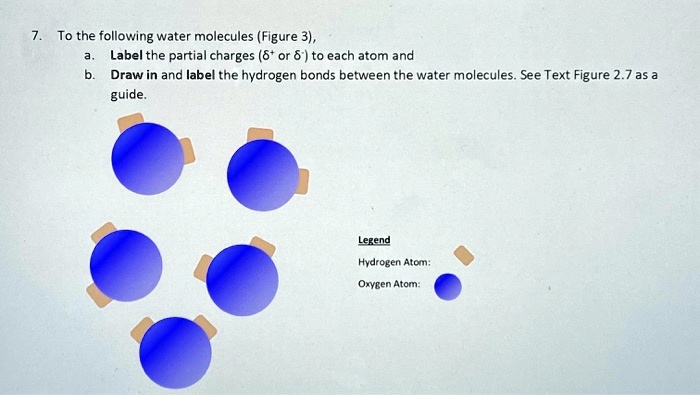
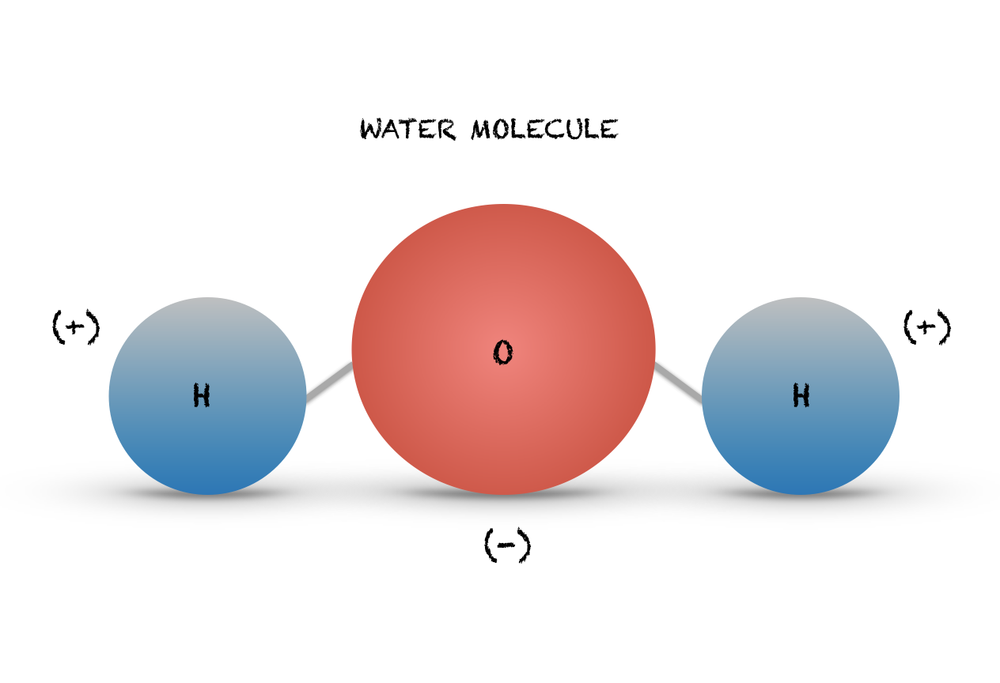
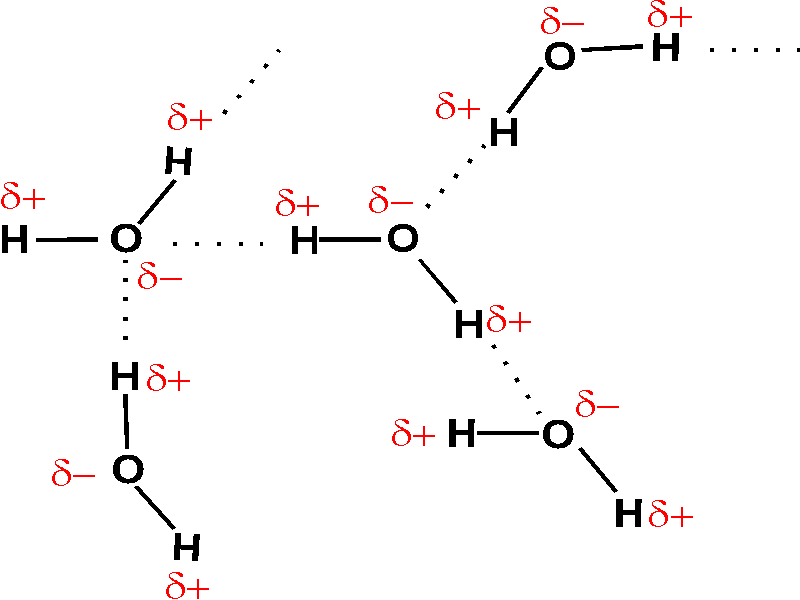
2.2 Water - Concepts of Biology - 1st Canadian Edition The hydrogen and oxygen atoms within water molecules form polar covalent bonds. The shared electrons spend more time associated with the oxygen atom than they do with hydrogen atoms. There is no overall charge to a water molecule, but there is a slight positive charge on each hydrogen atom and a slight negative charge on the oxygen atom. Structure of Water Molecule | Properties of Water 2023 - Botnam The structure of water molecule is made up of one molecule of oxygen and two molecules of hydrogen-bonded covalently. Water (H2O) is essentially considered one of the most important substances found on the earth. It covers over 70% of the earth's surface and makes up as much as 95% of the living organisms. The Structure of Water Molecule 1- Draw one water molecule. Be sure to label the atoms present ... 1- Draw one water molecule. Be sure to label the atoms present show the bonds present between the atoms show the shape of the molecule define any symbols ... The diagram shows hydrogen bonds between water molecules. Label the ... Answer: First square = [-] Second square = [+] Third square = H Forth square = O Fifth square = H Explanation: Advertisement batolisis Labeling the diagram to show how hydrogen bonds form between water molecules: 1st box = ( - ) 2nd box = ( + ) 3rd box = ( H ) 4th box = ( O ) 5th box = ( H )
Acid-base reaction - Dissociation of molecular acids in water The equation for the dissociation of acetic acid, for example, is CH 3 CO 2 H + H 2 O ⇄ CH 3 CO 2− + H 3 O +. Dissociation of bases in water In this case, the water molecule acts as an acid and adds a proton to the base. An example, using ammonia as the base, is H 2 O + NH 3 ⇄ OH − + NH 4+.
water - Covalent Bond/Hydrogen Bond - Chemistry Stack Exchange The covalent bond is the electrostatic interaction between two atoms in the same molecule. Covalent bonds are much stronger than hydrogen bonds: the $\ce {O-H}$ has a strength of 467 kJ/mol, while the hydrogen bond is usually between 4 to 40 kJ/mol. Just want to point out that there is a subtlety to the word "water":
Label Water Molecule Diagram | Quizlet Label Water Molecule 3.0 (4 reviews) + − Flashcards Learn Test Match Created by Jacqueline_Curls Terms in this set (5) hydrogen bond weak charge BETWEEN water molecules polar covalent bond Strong bond WITHIN one water molecule when electrons are UNEQUALLY shared. Hydrogen
15.1: Structure of Water - Chemistry LibreTexts Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent ( polar bonds ). The oxygen atom attracts the shared electrons of the covalent bonds to a significantly greater extent than the hydrogen atoms.
Draw and label a diagram of a water molecule below Make sure to ... In the case of water , the partially positive charged hydrogen atoms of the water molecule with interact with the negative charged oxygen molecule of another ...
H2O Molecular Geometry, Lewis Structure, Shape and Bond Angles The molecular geometry of any molecule depends on its Lewis structure, the arrangement of atoms, and its electrons. In an H2O molecule, the Oxygen atom forms two single sigma bonds with Hydrogen atoms. Although these two Hydrogen atoms are arranged symmetrically in the plane, the two lone pairs of electrons on the Oxygen atom push these atoms.
Hydrogen bonding in water (video) | Khan Academy To find out how many moles of water in 3 cups just divide 720 by 18.0015 which gives you 39.9667 moles of water. This gives you 240.685 * 10^32 or 2.40685 * 10^34 water molecules. Since there are 3 atoms in a water molecule multiply the number of water molecules by 3 giving you 7.2206 * 10^34 atoms. ( 6 votes)
Water | Definition, Chemical Formula, Structure, Molecule, & Facts The water molecule is composed of two hydrogen atoms, each linked by a single chemical bond to an oxygen atom. Most hydrogen atoms have a nucleus consisting solely of a proton. Two isotopic forms, deuterium and tritium, in which the atomic nuclei also contain one and two neutrons, respectively, are found to a small degree in water.
How do you label a water molecule? - Answers A Water Molecule has molecular 4mula "H2O". H+ & OH- are the charges on water.because of these charges,water molecule is called a polar molecule.
The Molecular Formula for Water - ThoughtCo The molecular formula for water is H 2 O. One molecule of water consists of one oxygen atom covalently bonded to two hydrogen atoms. The same formula is also the simplest formula for water, since the subscripts don't reduce to smaller values. There are three isotopes of hydrogen.
3.1.4 Draw and label a diagram showing the structure of - Studylib Many of water's properties depend on the structure of water molecules. Water molecules are charged, with the oxygen atom being slightly negative (δ-) and the ...
7.3: Hydrogen-Bonding and Water - Chemistry LibreTexts In water, each hydrogen nucleus is covalently bound to the central oxygen atom by a pair of electrons that are shared between them. In H 2 O, only two of the six outer-shell electrons of oxygen are used for this purpose, leaving four electrons which are organized into two non-bonding pairs.
Water Structure & Properties | Molecule & Physical Properties | A Level Structure of Water molecule. In a water molecule, each hydrogen atom shares an electron pair with the oxygen atom. The geometry of the water molecule is dictated by the shapes of the outer electron orbitals of the oxygen atom, which are similar to the bonding orbitals of carbon. These orbitals describe a rough tetrahedron, with a hydrogen atom ...
Water Molecule Structure & Diagram - Study.com Water is made of three atoms, an oxygen atom and two hydrogen atoms, and thus is both a molecule and a compound. A chemical formula is an abbreviation that shows which atoms are in the...
Water | H2O - PubChem Water | H2O | CID 962 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards ...
PDF CHEMISTRY OF WATER WORKSHEET - Loudoun County Public Schools The drawing here represents a water molecule. Label the oxygen and hydrogen atoms in this molecule: 2. You should have some water molecules on your table. ... In the water molecule below, shade in the area(s) where the electrons will spend more of the time. 8. The oxygen atom will have a partial charge on it. This will be a (positive or ...
Draw and label a ball and stick presentation of a water molecule ... This visual representation demonstrates that one atom of oxygen, O, bonds with two atoms of hydrogen, H, to form one molecule of water, ...
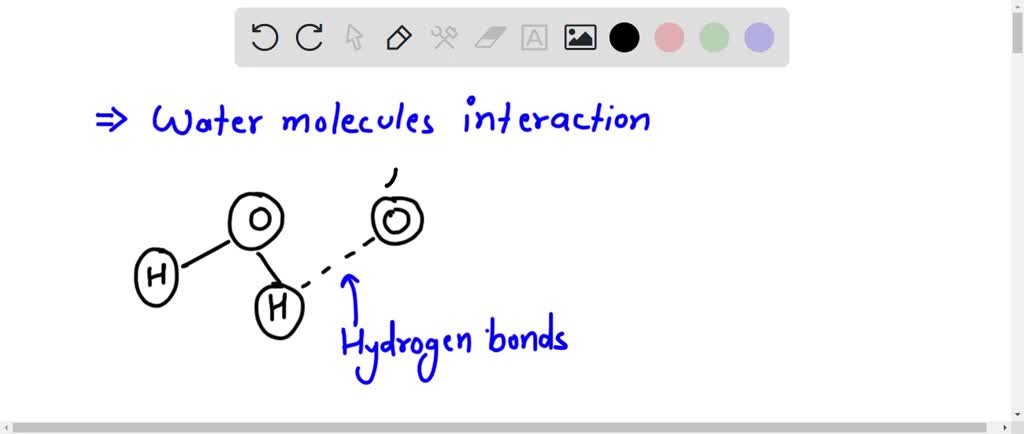
Picture below showing four water molecules interacting via hydrogen bonds? be sure lo label partial charges in each molecule and use dotted line to represent a hydrogen bond.
Draw a diagram of water molecules, labeling the hydrogen bond and ... A diagram of water molecules, labeling the hydrogen bond and covalent bond - is attached as an image. Water has hydrogen and oxygen molecules bonded with covalent and, covalent bonds are intramolecular bonds whereas hydrogen bonds are intermolecular bonds. Water is held together with covalent bonds.
The Structure of Water | Chemistry for Non-Majors | | Course Hero Structure of Water. Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent ( polar bonds ). The oxygen atom attracts the shared electrons of the covalent bonds to a significantly greater extent than the hydrogen atoms.
















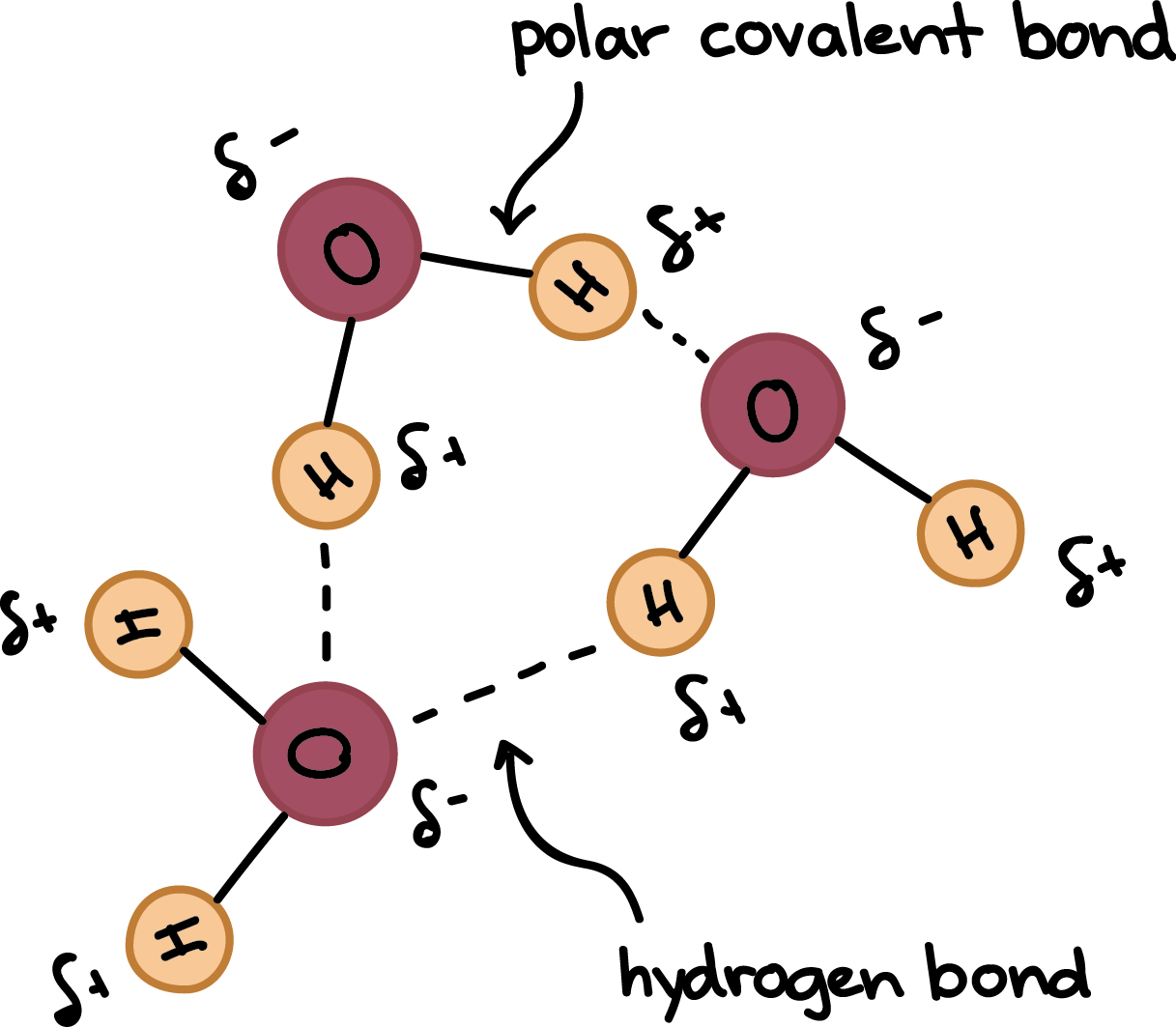
Post a Comment for "38 label the water molecule"